
Regulatory bodies are paying increasing attention to hospital and health system pharmacies regarding sterile medication compounding. Enforcement of USP <797> and <800> revisions and regulations are slated to take effect on December 1, 2019, increasing pressure to be in compliance.
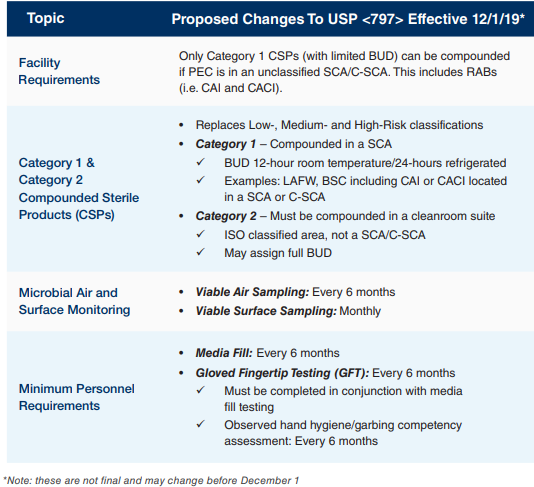
In preparation for the December 1 deadline, a hospital system chief pharmacy officer, who previously partnered with Comprehensive Pharmacy Services (CPS), brought CPS in to do an initial assessment and evaluate USP <797> and <800> compliance across ten pharmacies in hospitals and infusion centers across a large hospital system.
CPS assigned a sterile compounding expert to spend one day at each of the ten sites reviewing USP <797> and <800> standards in the following areas:
• Physical plant and equipment
• Work practices
• Competencies and training
• Environmental monitoring
• Quality assurance activities and documentation
Because CPS sterile compounding experts are pharmacists who have worked in hospital and health system pharmacies, they take a holistic approach to the issues that directors of pharmacies and their staff face. The director of pharmacy can assign a staff pharmacist or lead technician to liaison with the CPS sterile compounding expert.
“Because we’ve done so many of these assessments in so many hospitals and large health systems, we’ve become very proficient and can get information very quickly. In addition, we take a non-invasive and collaborative approach to conducting assessments that minimally interrupts the work flow, while being educational to staff,” said Chris Beebe, Division Vice President, Medication Safety for CPS.
Following the assessments, each site received a full report and action plan that identified areas of non-compliance or partial compliance along with strategies to correct those areas.
PHASE 2: Continuous Readiness Implementation
The action plans identified multiple opportunities for improvement and standardization across the ten sites. The chief pharmacy officer recognized the need for a single point of contact to coordinate implementing the recommended changes and standardization across the hospitals and infusion centers and selected the CPS expert to function as that single point of contact. The implementation phase is currently an ongoing effort encompassing three years in order to ensure compliance to meet the December 1 deadline and beyond. During the implementation phase, the CPS sterile compounding expert is on-site three days every month to:
• Track progress on the action plan at each location
• Work with corporate pharmacy leadership to implement standardization throughout the hospital system
• Interface with architects and engineers tasked with building new pharmacies and/ or making changes to existing pharmacies to ensure that the construction of physical space is in compliance with USP standards
To standardize processes, the CPS expert works with system departments:
• Environmental services and housekeeping leadership to implement a standardized IV room cleaning program across the enterprise
• Infection prevention department to standardize environmental monitoring program
• Regulatory affairs department to assist with survey preparedness related to USP
• Education department to standardize pharmacy competency training
The CPS sterile compounding expert also leads the system hazardous drug task force to implement strategies related to USP <800> compliance covering assessment of risk, preparation and administration of hazardous drugs, and education of all staff involved in the process. The CPS expert is on tap for support if the system has an accreditation survey issue related to USP during the continuous readiness engagement. By participating in the continuous readiness program, the system also has access to the CPS sterile compounding toolkit which typically is only available to CPS managed sites. “Our toolkit includes a standardized assessment of risk for hazardous drugs on a hospital’s formulary,” said Beebe. “It’s an important tool for hospitals and was developed to be a comprehensive yet straightforward method to assess, document, and communicate the containment strategies a hospital will employ for these drugs.”
Following the initial sterile compounding assessments and ongoing continuous readiness process including implementation of the action plans, subsequent regulatory surveys at system sites resulted in no USP-related findings.

As the USP enforcement date approaches, hospitals and health systems must prepare
to ensure that they are as compliant as possible. CPS brings a breadth of experience in
hospitals of all types and sizes. Because they understand what surveyors are looking for,
they have the expertise to set client hospitals and health systems up with a continuous
readiness program that ensures their success.